Oxygen Atom Drawing
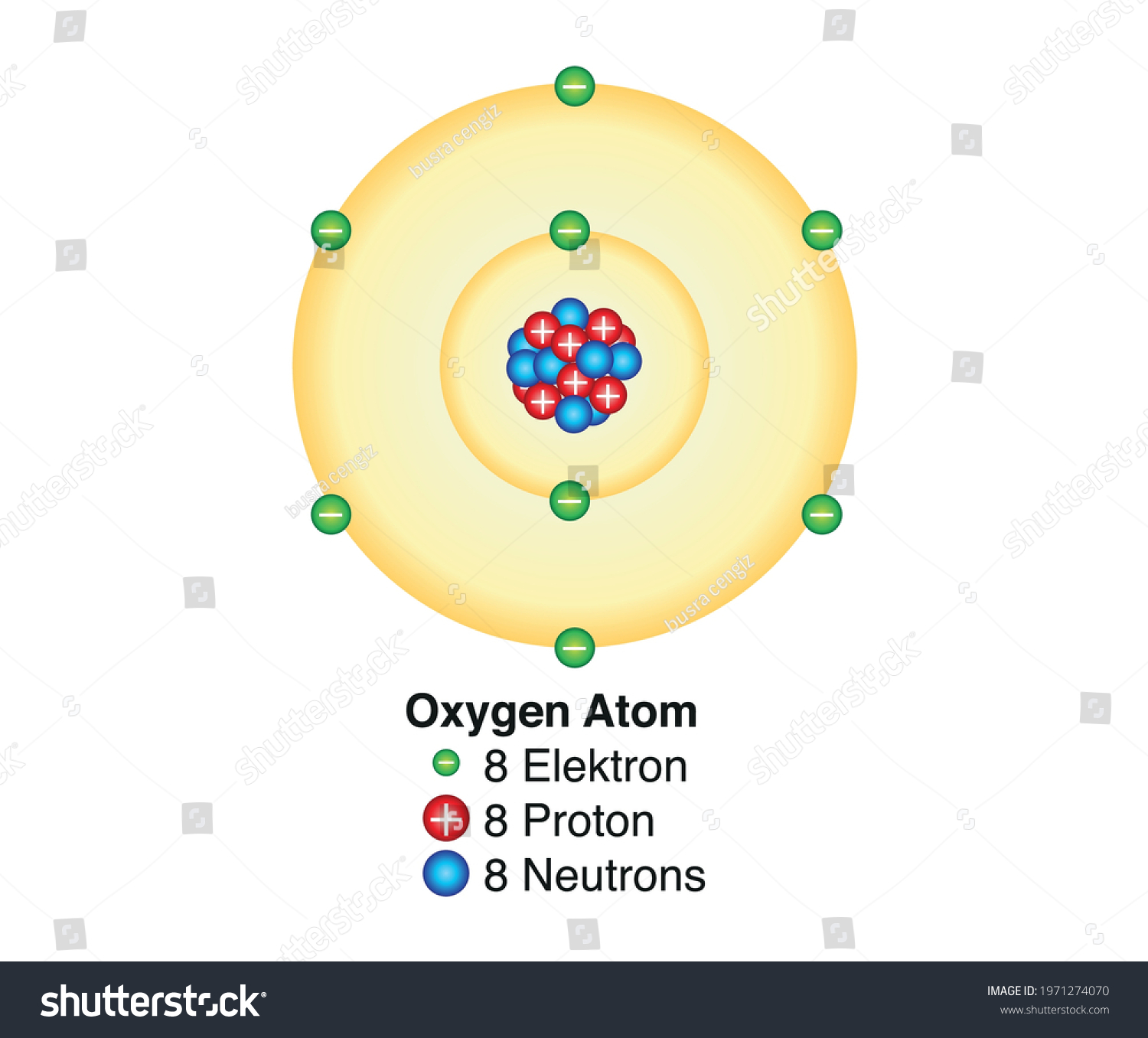
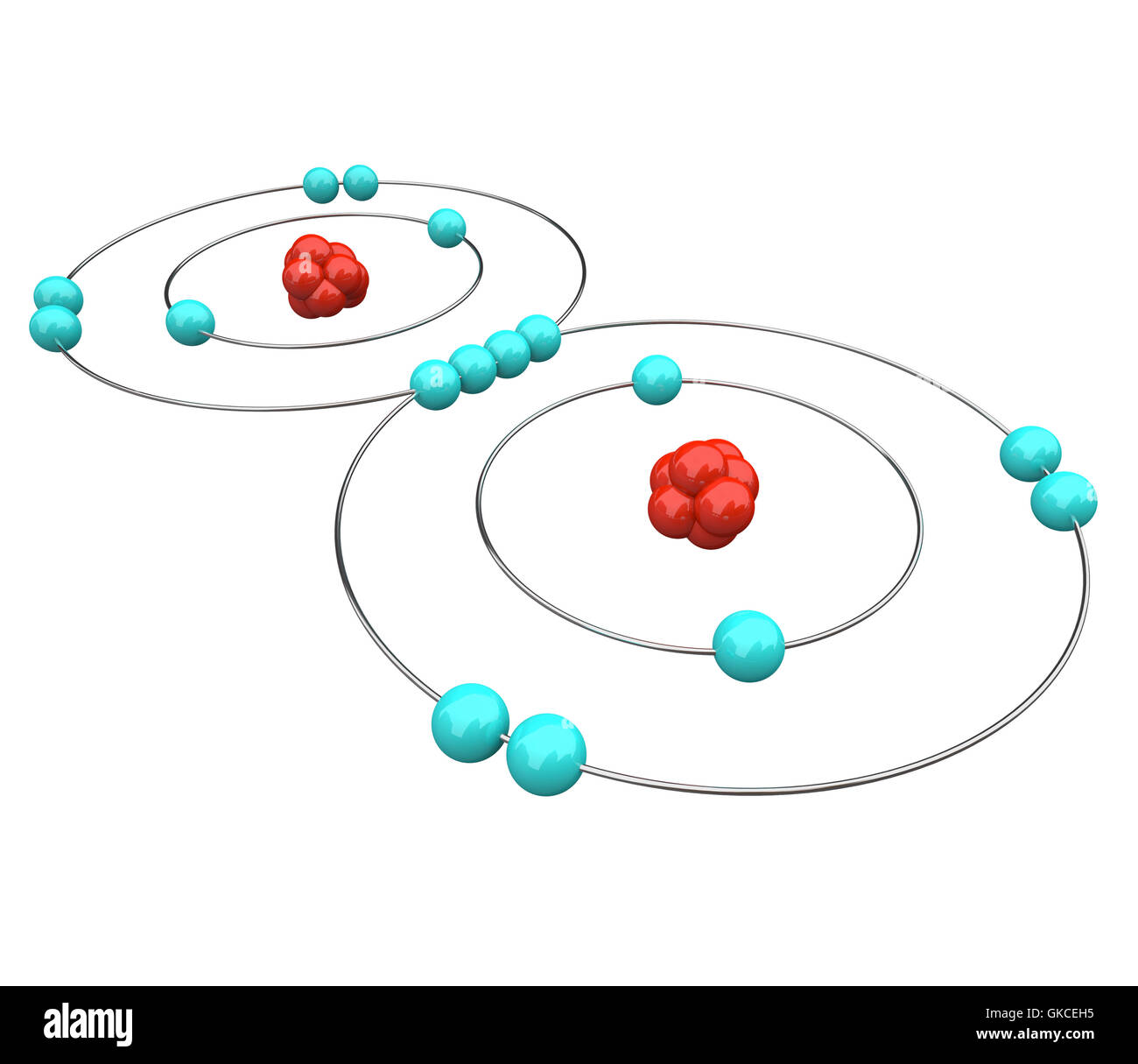
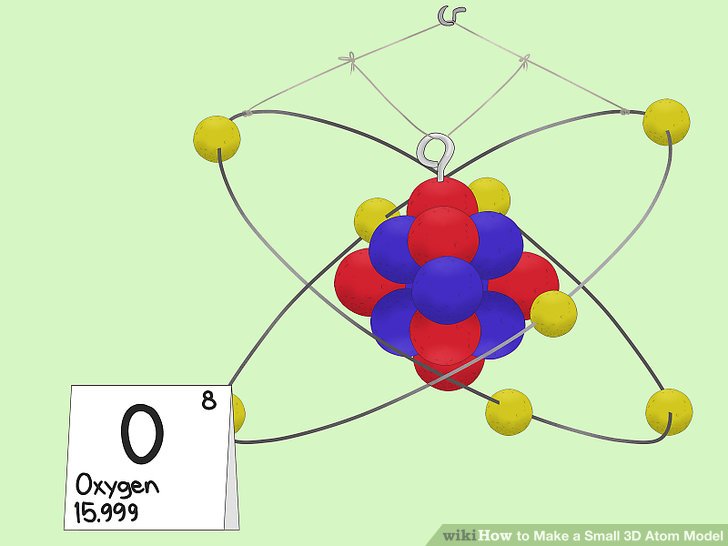
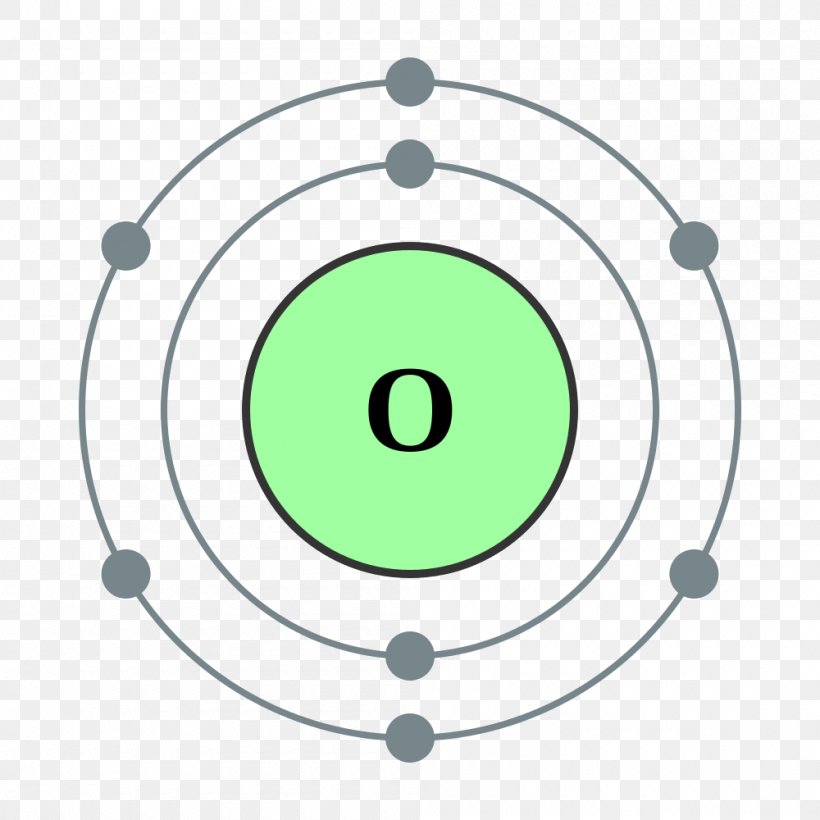
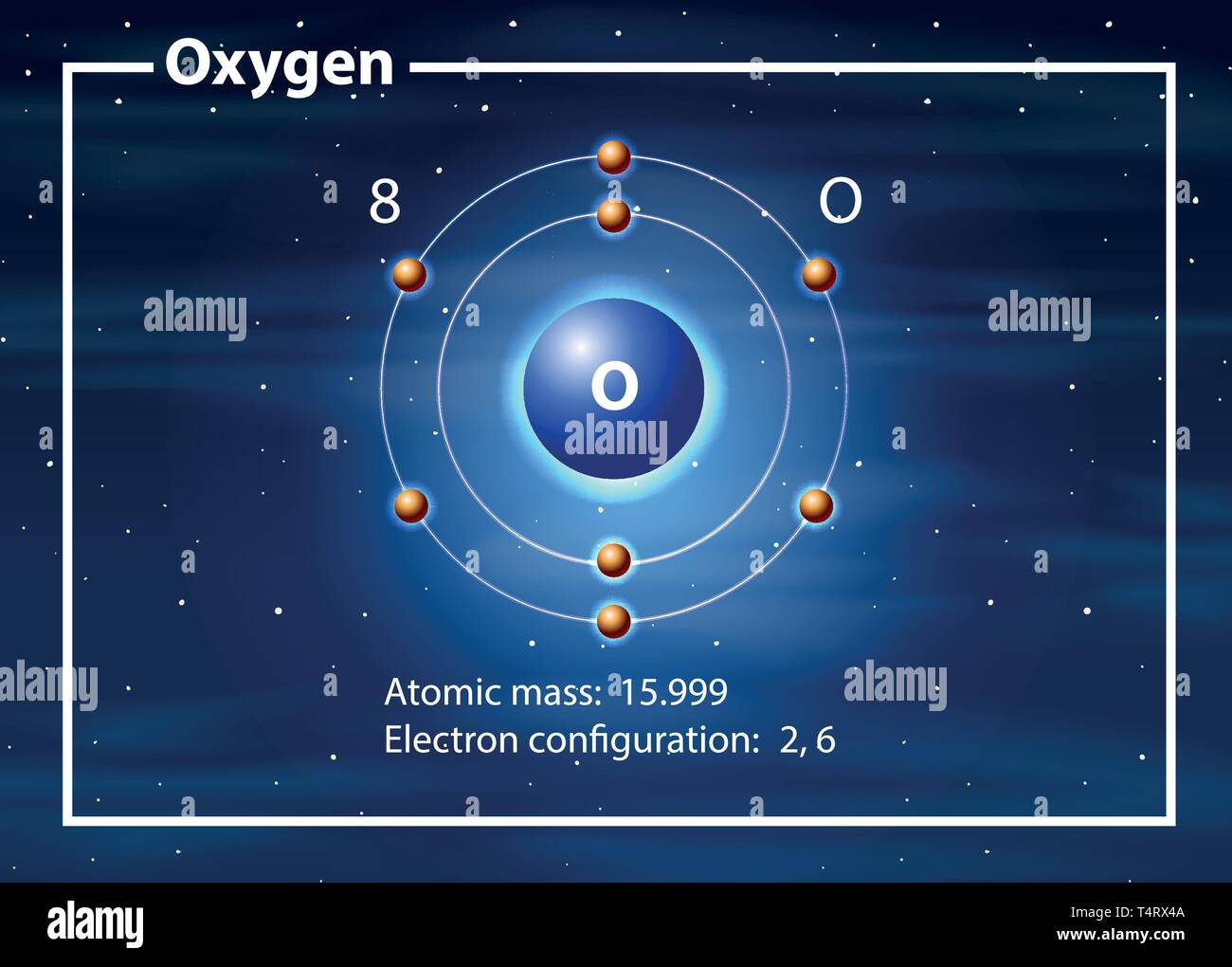
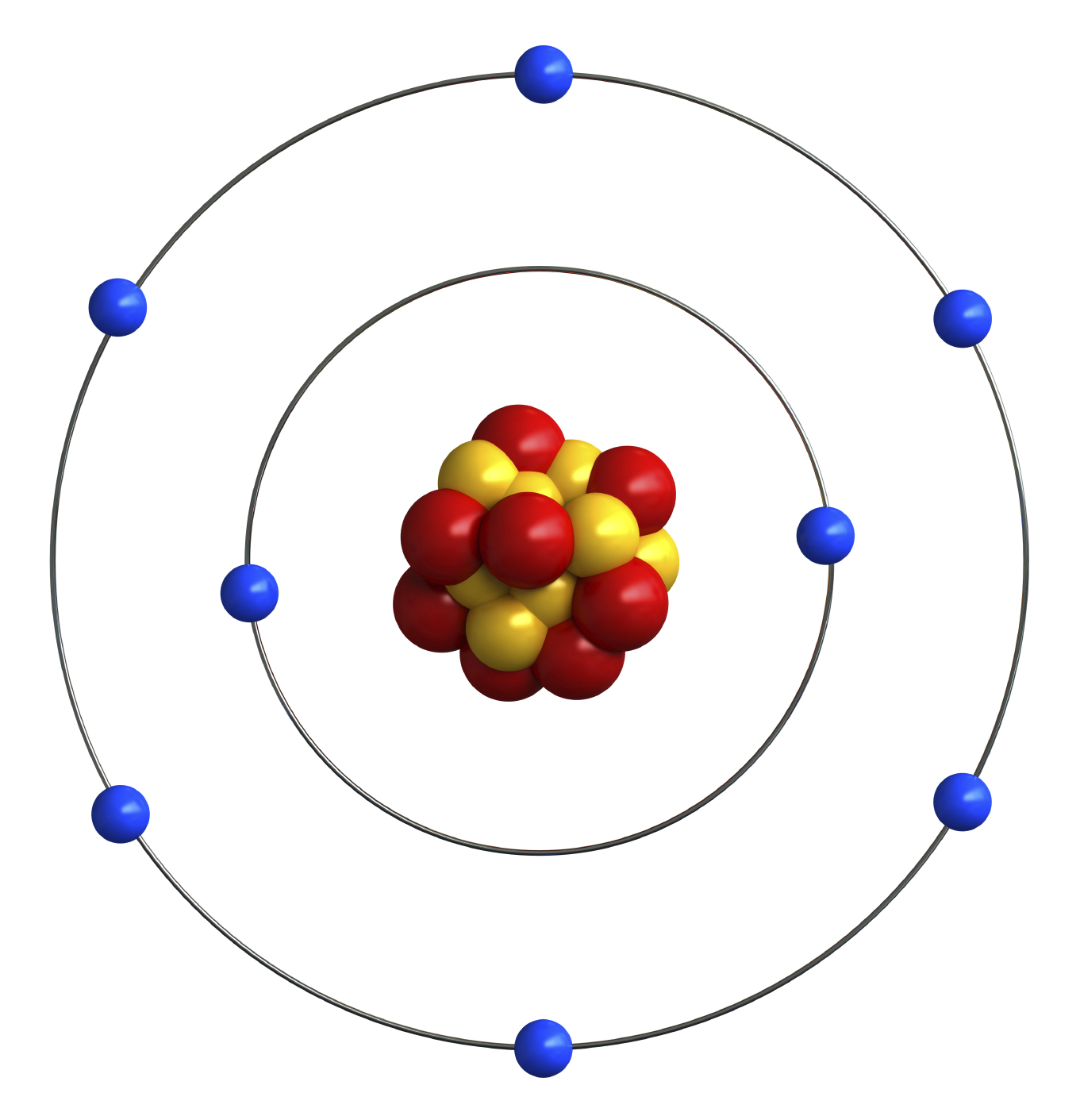
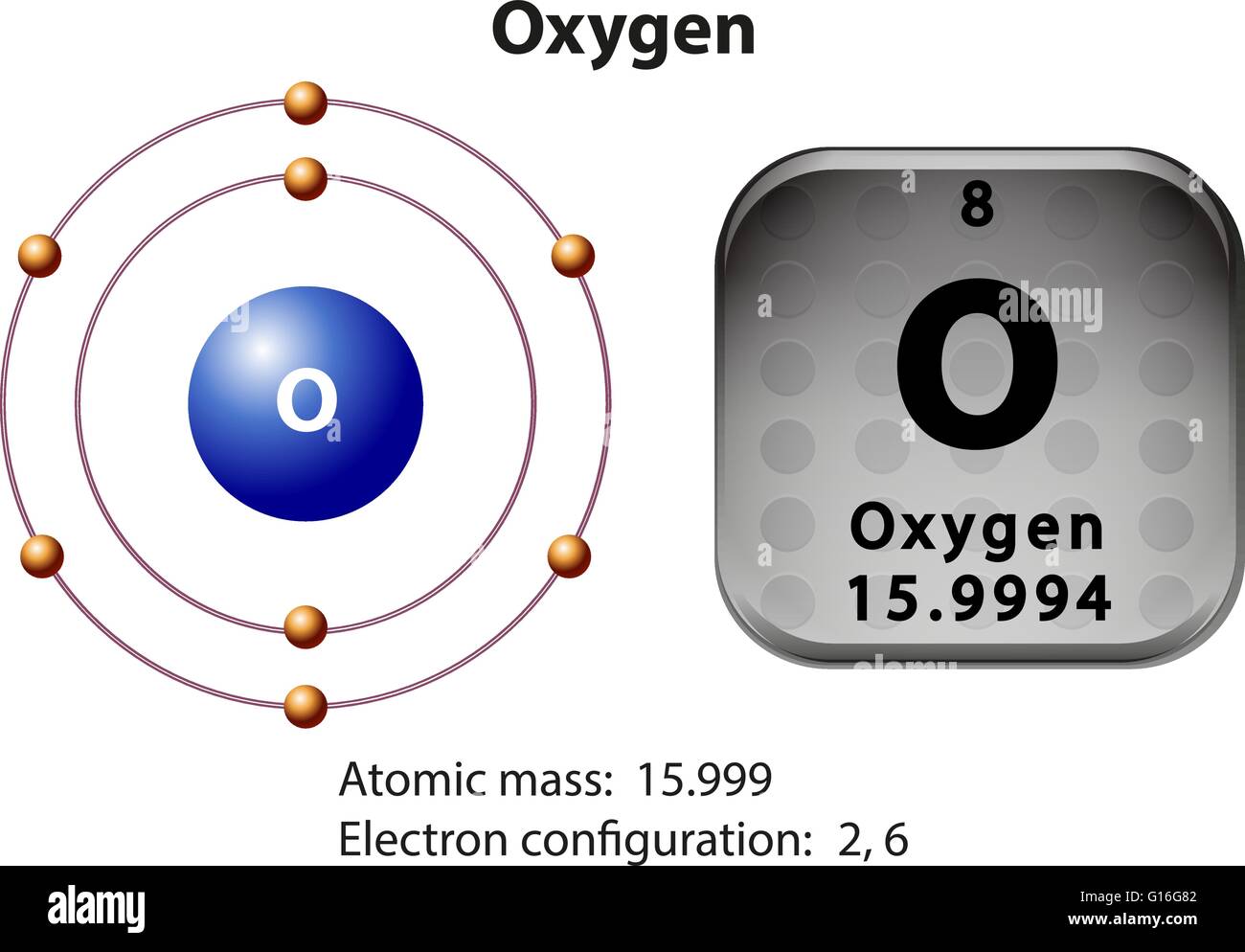
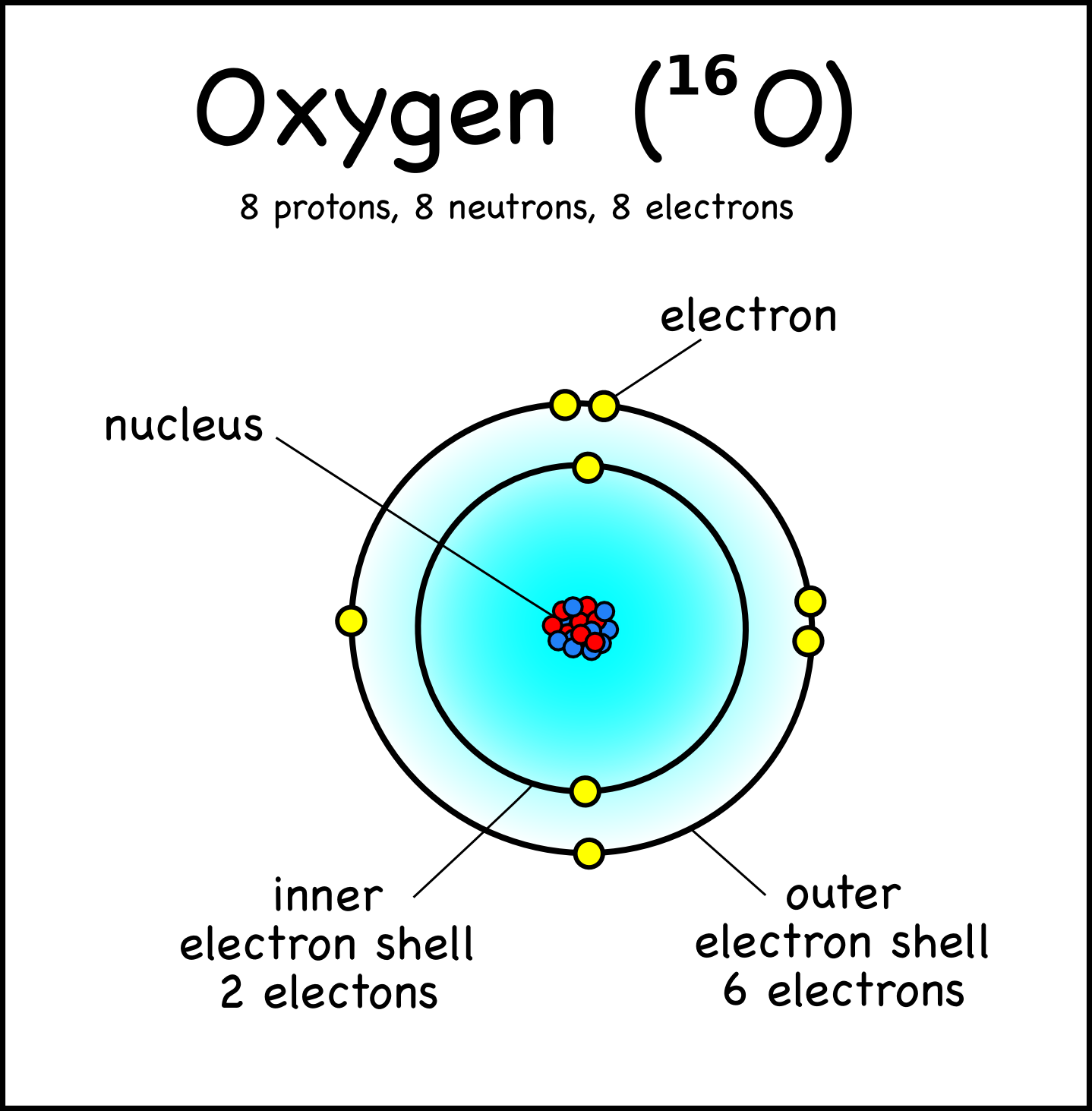
Oxygen Atom Drawing - Oxygen is neutral and its atomic number is 8, hence, the number of protons and electrons available for its bohr diagram is also 8. Web a diagram of an oxygen atom. The k shell contains 2 electrons and the m shell contains 6 electrons. Web in this video we'll look at the atomic structure and bohr model for the oxygen atom (o). Web the dot diagram for oxygen is a visual representation that allows us to understand the electron configuration of an oxygen atom. Chemists normally represent a bond using a line instead of two dots. In the bohr model, electrons are pictured as traveling in circles at different shells,. Electrons cannot exist at energies between these levels. Web so if we ignore hydrogen, the central atom is either going to be carbon or oxygen. For that, we have electron shell diagrams. Web learn about the structure of an oxygen atom with our diagram and understand the key components, including protons, electrons, and neutrons. The energy levels in a bohr model are also called shells. Web the number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons); So that they’d have a bit of context, i went over the basic parts of an atom (protons, neutrons, and electrons) and made it clear that the name of the element is determined solely by the number of protons. Web in a water molecule, an oxygen atom forms two bonds, one to each hydrogen atom. The diagram would show the nucleus at the center, surrounded by two electron shells. Start by illustrating the nucleus, and then draw the two electron shells. This diagram shows how the electrons in the oxygen atom are arranged in different orbitals. Sources, facts, uses, scarcity (sri), podcasts, alchemical symbols, videos and images. Web the rings in a bohr model represent the discrete energy levels that electrons can occupy. Start by illustrating the nucleus, and then draw the two electron shells. The shells are labelled as shown for the chlorine model below. It's easier to understand electron configuration and valence if you can actually see the electrons surrounding atoms. The first shell should contain 2 electrons, and the second holds the remaining 6 electrons. Web learn about the structure. Electrons cannot exist at energies between these levels. This is especially true of the nonmetals of the second period of the periodic table (c, n, o, and f). The structures of h 2 , f 2 , and h 2 o would usually be drawn as follows: The k shell contains 2 electrons and the m shell contains 6 electrons.. Start by illustrating the nucleus, and then draw the two electron shells. To do that we need to find the number of electrons for the oxygen atom. Web the bohr model of oxygen is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 6 electrons. Web rutherford’s gold foil experiment. A bohr. The energy levels in a bohr model are also called shells. Start by illustrating the nucleus, and then draw the two electron shells. The shells are labelled as shown for the chlorine model below. I show you where oxygen is on the periodic table and how to determine how many valence electrons it has. Web rutherford’s gold foil experiment. Hence, the oxygen atom has 6 valence electrons. I show you where oxygen is on the periodic table and how to determine how many valence electrons it has. Web the bohr model of oxygen states that the nucleus of the oxygen atom contains 8 protons and 8 neutrons, while 8 electrons revolve around the nucleus in 2 energy levels. Electrons. Orbital is the region of space around the nucleus of an atom where electrons are found. Web rutherford’s gold foil experiment. Web the number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons); So that they’d have a bit of context, i went over the. Memorization over the winter break. Web in this video we'll look at the atomic structure and bohr model for the oxygen atom (o). Oxygen is more electronegative than carbon. Web the dot diagram for oxygen is a visual representation that allows us to understand the electron configuration of an oxygen atom. Learn for free about math, art, computer programming, economics,. Web the rings in a bohr model represent the discrete energy levels that electrons can occupy. Hence, the oxygen atom has 6 valence electrons. Ca), in their correct order — by atomic number — over their winter break. Oxygen is more electronegative than carbon. Web the dot diagram for oxygen is a visual representation that allows us to understand the. And let's look at our periodic table to figure out the relative values for those. Web in a water molecule, an oxygen atom forms two bonds, one to each hydrogen atom. We’ll use a bohr diagram to visually represent where the electrons are around the nucleus of the oatom. Web to draw the oxygen bohr model, outline the 8 protons,. Web updated on november 05, 2019. Start by illustrating the nucleus, and then draw the two electron shells. We’ll use a bohr diagram to visually represent where the electrons are around the nucleus of the oatom. The first shell should contain 2 electrons, and the second holds the remaining 6 electrons. 2 electrons can go in the first shell, 8. The first shell should contain 2 electrons, and the second holds the remaining 6 electrons. This is especially true of the nonmetals of the second period of the periodic table (c, n, o, and f). Web the first step, however, is to teach them how to draw basic models of atoms. Orbital is the region of space around the nucleus of an atom where electrons are found. For that, we have electron shell diagrams. Web in a water molecule, an oxygen atom forms two bonds, one to each hydrogen atom. How many valence electrons does the oxygen atom have? A bohr model of an oxygen atom is shown below. Hence, the oxygen atom has 6 valence electrons. We’ll use a bohr diagram to visually represent where the electrons are around the nucleus of the oatom. In the bohr model, electrons are pictured as traveling in circles at different shells,. So that they’d have a bit of context, i went over the basic parts of an atom (protons, neutrons, and electrons) and made it clear that the name of the element is determined solely by the number of protons. Explore the atomic model and discover the importance of oxygen in various chemical reactions and biological processes. Web the number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons); The structures of h 2 , f 2 , and h 2 o would usually be drawn as follows: The energy levels in a bohr model are also called shells.Basic Model Oxygen Atom Containing Protons vetor stock (livre de
Oxygen Atomic Diagram Stock Photo Alamy
Oxygen Atom Drawing at GetDrawings Free download
Bohr Model Oxygen Chemical Element Atomic Number, PNG, 1000x1000px
Oxygen atom diagram concept illustration Stock Vector Image & Art Alamy
Oxygen Atom Diagram Concept Stock Vector Illustration of research
Forms of Energy ND Studies Energy Curriculum
Symbol and electron diagram for Oxygen illustration Stock Vector Image
Molecular Structure of an Oxygen Atom Stock Vector Illustration of
Drawing Atoms Montessori Muddle
Atomic Number, Mass Number, And Isotopes.
It's Easier To Understand Electron Configuration And Valence If You Can Actually See The Electrons Surrounding Atoms.
Web The Bohr Model Of Oxygen States That The Nucleus Of The Oxygen Atom Contains 8 Protons And 8 Neutrons, While 8 Electrons Revolve Around The Nucleus In 2 Energy Levels.
Chemists Normally Represent A Bond Using A Line Instead Of Two Dots.
Related Post: